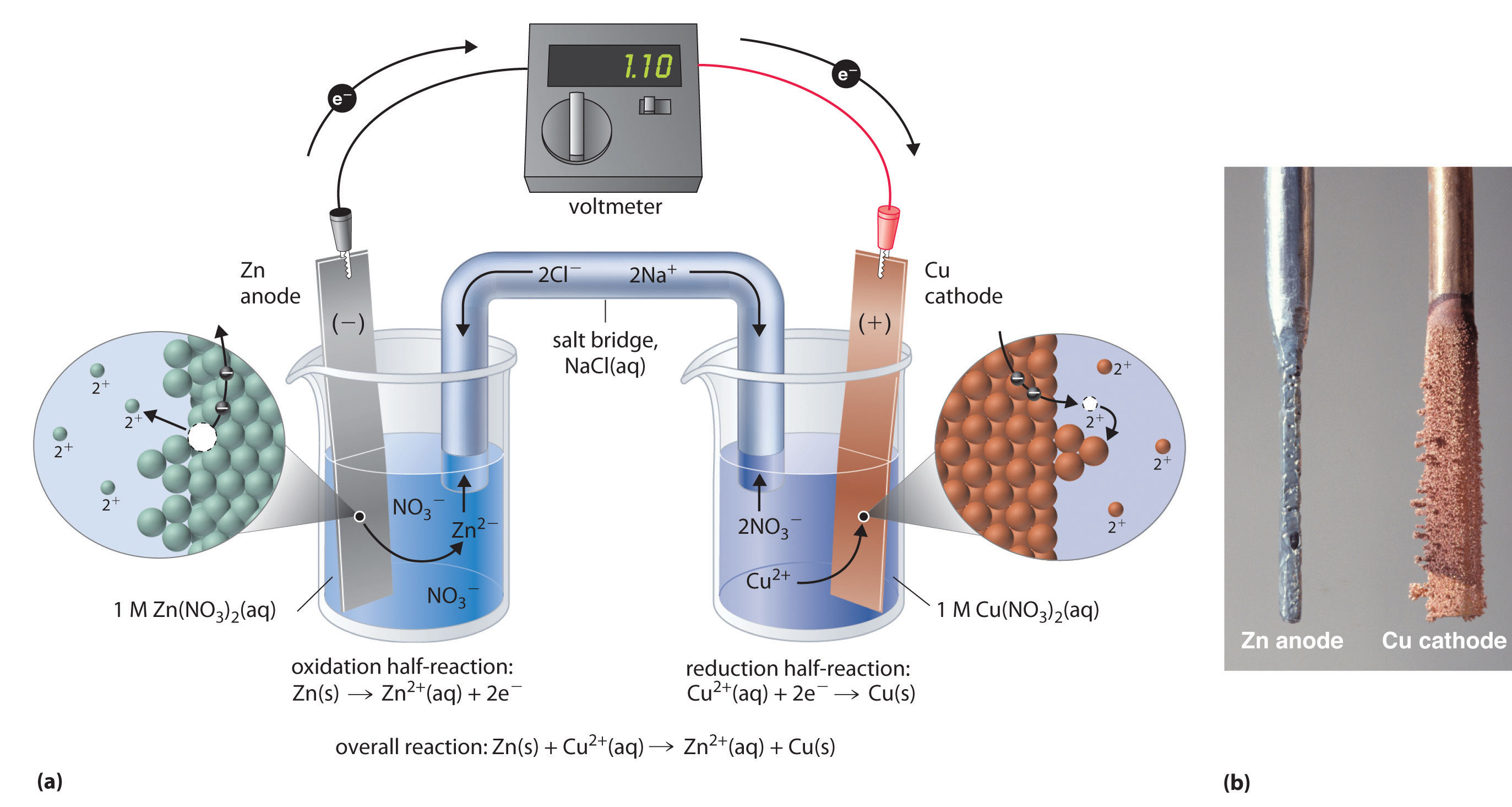
Only the gamma form of manganese dioxide performs well as an active cathode material in alkaline. This battery chemical reaction, this flow of electrons through the wire, is electricity – ready to power everything from your digital camera and handheld game, to your wireless mouse, flashlight and portable clock. The primary alkaline battery is normally cathode limited. When the circuit is closed, the stronger attraction for the electrons by the manganese dioxide will pull the electrons from the zinc anode electrode through the wire in the circuit to the cathode electrode. This happens when the battery is placed in a device and the device is turned on – same principle as turning on and off a light switch in your house. 
The alkaline electrolyte is used as either potassium or sodium hydroxide. The battery is called an alkaline battery because of its. The stored electrons will only flow when the circuit is closed. The alkaline battery mainly consists of zinc and manganese dioxide as electrodes. In Alkaline batteries, the cathode is made of Manganese Dioxide and the anode is made of Zinc. In simple terms, each battery is designed to keep the cathode and anode separated to prevent a reaction. An alkaline battery produces electricity when the manganese dioxide cathode is reduced and the zinc anode becomes oxidized. 3 A separator that prevents contact between the anode and. 2 The anode (the negative side), commonly made out of graphite, the same material found in many pencils. It has four key parts: 1 The cathode (the positive side), typically a combination of nickel, manganese, and cobalt oxides. This leads electrons moving towards the cathode, creating a voltage potential between the cathode and the anode. When charging, a buildup of positive ions forms at cathode/electrolyte interface. The efficiency of the reaction depends on the quality of the raw materials and availability of water and hydroxyl ions during reaction. A lithium-ion battery is a type of rechargeable battery. An electrochemical battery consists of a cathode, an anode and electrolyte that act as a catalyst. Energizer ® Performance Metal Tactical LightsĪpplying this battery chemistry to the real world, the electrons (e) generated during the reaction are used to power devices.Energizer ® Vision HD Performance Metal.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
